 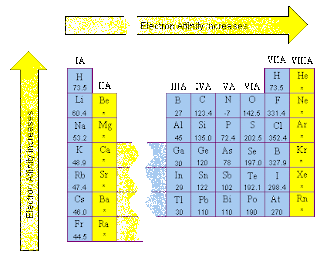
The first electron affinities of the group 7 elements F By convention, the negative sign shows a release of energy. For example, the first electron affinity of chlorine is -349 kJ mol -1. It is the energy released (per mole of X) when this change happens.įirst electron affinities have negative values. This is more easily seen in symbol terms. The first electron affinity is the energy released when 1 mole of gaseous atoms each acquire an electron to form 1 mole of gaseous 1- ions. Electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 6 and 7 of the Periodic Table. Ionisation energies are always concerned with the formation of positive ions. Important! If you aren't reasonable happy about atomic orbitals and electronic structures you should follow these links before you go any further. It assumes that you know about simple atomic orbitals, and can write electronic structures for simple atoms. This means that Li will display greater electron affinity than Na.This page explains what electron affinity is, and then looks at the factors that affect its size. Hence, the attraction between the Li nucleus and any potentially accepted electron will be greater resulting in a greater electron accepting capacity (affinity) for Li. So, the new electron accepted by Li will enter its valence shell and will be closer to its positively charged nucleus compared to the electron added to the valence shell of Na. Here, the explanation is similar as well. For this trend, an increase is observed across a period, from left to right, and a decrease is observed as we go down a group. In other words it represents the change in energy occurring when an electron is gained by an atom. So, overall, Li has a higher ionization energy than Na.Finally, electron affinity refers to an atom's ability to accept electrons. This means that less energy will be required in order to remove any Na electrons. In the case of Na, the valence electrons are in the 3rd energy level, so they are not located that close to the nucleus and hence they are less attracted by it. As a consequence more energy is required so that to overcome this attraction and remove an electron (as the first electrons of an atom removed are those further away from the nucleus, because they are the ones less attracted by it). For example, for Li, the valence electrons are placed to the 2nd energy level which is closer to the nucleus meaning that they are more attracted by it. In this case the trend increases across a period, from left to right, whereas it decreases down a group (same as with electronegativity). More accurately, ionization energy represents the amount of energy required for the removal of one or more electrons from an atom. Hence, Li will display greater electronegativity than Na.Ionization energy is often quoted as the opposite of electronegativity. As previously mentioned, Li has a smaller atomic radius and therefore its nucleus is closer to its valence shell (and hence valence electrons) so it can exert a greater pulling force towards the attracted electrons compared to the nucleus of Na (where the valence shell is further away from the nucleus so the attractive force is less). For electronegativity, the trend that is observed is that it increases across a period, from left to right, and that it decreases as we go down a group. Therefore, overall, Na has a greater atomic radius than Li.Electronegativity is the chemical property/ability of an atom to attract electrons towards it, due to its positively charged nucleus, and form bonds. This means that in the Na atom, the electrons are further away from the nucleus and therefore less attracted by it, resulting in the atom occupying more space and therefore its atomic size being greater. Respectively for the elements of Li and Na, Li is positioned in the 2nd period and has its valence electrons in the 2nd energy levels whereas Na belongs to the 3rd period and hence has its valence electrons placed in the 3rd energy level. In the periodic table, across a period, from left to right, the atomic radius decreases whereas down a group it increases. More specifically, it refers to the half-distance between the two nuclei of two adjacent atoms of the same element. The Atomic radius of an atom represents its atomic size. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
